|
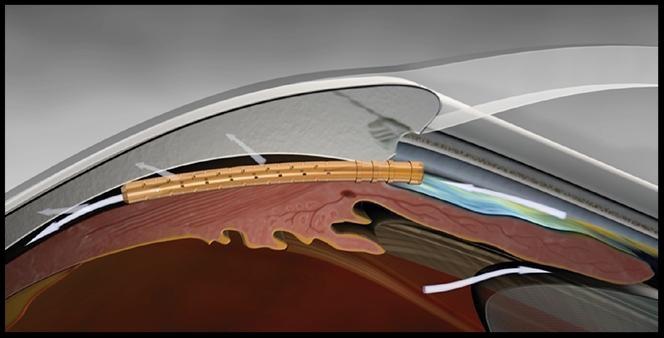
Introduction As you are probably aware, on August 29th, 2018, Alcon voluntarily withdrew the CyPass device due to safety concerns based on five-year data. The concerns centered around a higher rate of endothelial cell loss in patients receiving cataract surgery plus CyPass compared to cataract surgery alone. Overview of Results
At five years, patients undergoing cataract surgery with CyPass demonstrated a 20.5% loss of endothelial cells vs. 10% in the control or cataract surgery only group (of note, the control group was unusually low compared to other published data). There appears to be a correlation between CyPass implantation depth and rate of endothelial cell loss. For eyes with:
Of note, early migration of the CyPass device has been reported and, according to the Preliminary ASCRS CyPass Withdrawal Consensus Statement, "the potential for migration remains an important variable to be considered for long-term diagnostic monitoring". What does this mean and what should I do for my patients with CyPass? It's important to remember a few pertinent points. One, most of the endothelial cell loss did not occur until year four and five so it may be a while before we were to see any potential complications. Two, although certainly a risk factor, not everyone with endothelial cell loss will develop corneal edema. Three, removal or repositioning of a CyPass without the presence of corneal decompensation is likely to cause further endothelial cell loss. The current recommendations for monitoring patients should include:
Monitoring these patients with clinical examination at standard glaucoma follow ups should be adequate. We are always happy to monitor and evaluate patients as needed. Any evidence or concern for corneal decompensation should be referred back to the surgeon for further evaluation. Speaking of the CyPass withdrawal, VisionAmerica's glaucoma specialist, Dr. Karen Shelton noted, "For a minimally invasive procedure, CyPass provided a great response for many patients. I hated to see it withdrawn but understand the concerns. I don't anticipate having to remove or manipulate previously inserted devices unless we start to see corneal changes." If you have any questions or concerns, please don't hesitate to give us a call.
0 Comments
Leave a Reply. |
AuthorThe staff and doctors at VisionAmerica are committed to providing relevant information for you, your patients and your practice. We hope you find the information in our blog post helpful. Archives
August 2019
Categories |
 RSS Feed
RSS Feed
