|
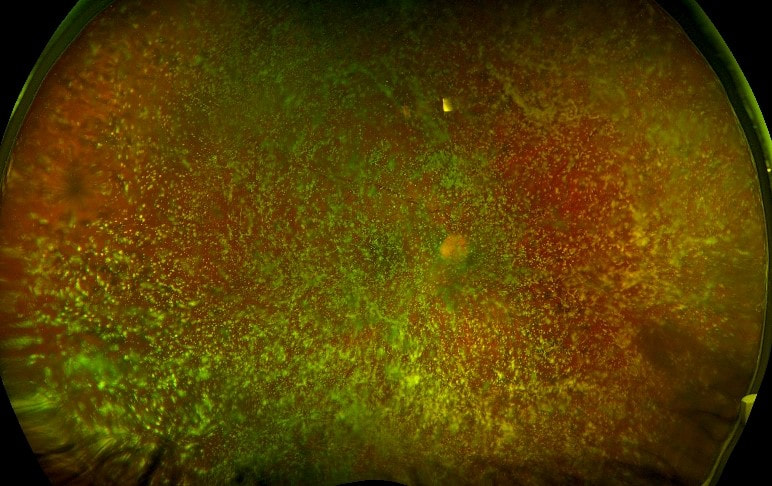
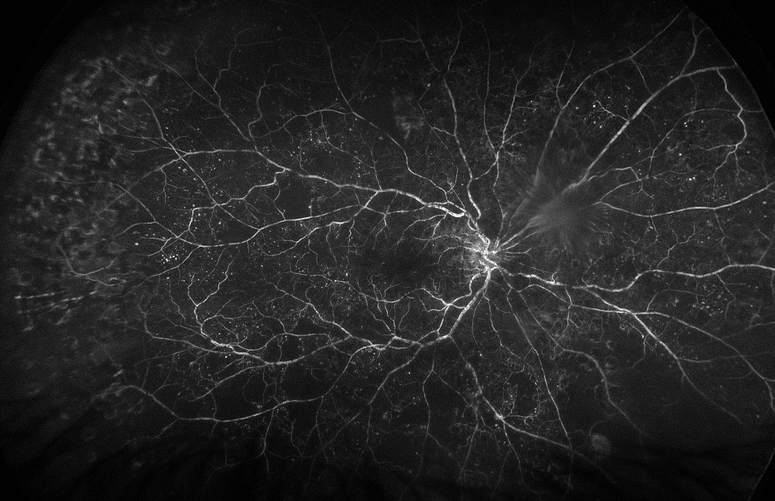
1/31/2018 0 Comments Photo Challenge of the Month!The two images below are from the right eye of the same patient.
The first image obviously shows extensive asteroid hyalosis. The second image is the fluorescein angiogram of the same eye on the same visit and shows proliferative diabetic retinopathy (which would have otherwise been missed on traditional fundus exam). Why is the angiogram able to provide such greater clarity than the traditional fundus photo? Click here to post your answer in our Facebook group! P.S. I had to look it up.
0 Comments
As the adoption of Minimally Invasive Glaucoma Surgeries (MIGS) like CyPass or iStint continues to grow, we thought it might be helpful to review the staging options for primary open angle glaucoma.
When referring a patient for a possible MIGS procedure, it is important to remember that most of these MIGS devices are only approved for use in patients with mild to moderate glaucoma at the time of cataract surgery. As such, visual field documentation is critical to support the medical necessity of these procedures. Below is a quick reference guide to choosing the appropriate stage. (Editor's Note: Gerriann Fagan spoke at our 2017 VisionAmerica Summer Conference and received rave reviews from the attendees. We are so excited to have her periodically contributing to our content!
If you enjoy the piece below, please reach out to her and let her know. You can learn more about Gerriann and the work she does at Warren Averett by clicking here!) We only have 24 hours in the day and 8,760 hours each year to manage our businesses, our families and our lives - not necessarily in that order. If you, or your team members, struggle to manage EVERYTHING, help is on the way. Making time for play means making time for focus, or does it? On Tuesday, December 20th, The U.S. Food and Drug Administration (FDA) approved LUXTURNA, what is hoped to be a one-time gene therapy product for patients with a confirmed biallelic RPE65 mutation-associated retinal dystrophy.
We first mentioned LUXTURNA in our email on Thursday, October 26th. Please click here if you missed it! LUXTURNA is for patients with mutations on both copies of the RPE65 gene who also have sufficient viable retinal cells to undergo therapy. According to a press release by the manufacturer of LUXTURNA, Spark Therapeutics, it "is the first FDA-approved gene therapy for a genetic disease, the first and only pharmacologic treatment for an inherited retinal disease (IRD) and the first adeno-associated virus (AAV) vector gene therapy approved in the U.S." ID Your IRD is a program provided by Spark Therapeutics which provides free genetic testing for a specific panel of inherited retinal dystrophies.
This program is undergoing significant change at the end of January. Any test kits submitted prior to January 31 will undergo testing of the established panel of the more common mutations associated with approximately 12 inherited rod-mediated retinal dystrophies. After January 31, they will only be processed as a single gene test for biallelic RPE65 mutation. This is the mutation targeted with their recent gene therapy approval, Luxterna. We will need to see any patients with inherited retinal dystrophies, who are looking for free testing options, immediately. While not comprehensive, this genetic testing panel is the only free option currently available to provide a molecular diagnosis for some of these patients. If you have any questions, please feel free to call or email Dr. Nowakowski. 205-943-4600 or [email protected] |
AuthorThe staff and doctors at VisionAmerica are committed to providing relevant information for you, your patients and your practice. We hope you find the information in our blog post helpful. Archives
August 2019
Categories |

 RSS Feed
RSS Feed
